
You will use these graduated cylinders for measuring volume. (Actually, the 25 mL graduated cylinder won't be used for the field lab this Spring.)

Portable Digital Balance:
You will need the portable (pocket) digital balance for measuring mass.
For this lab you will be measuring mass in grams, so be sure your balance has a small "g" above the "0.00". If not, use the MODE button to show the "g".

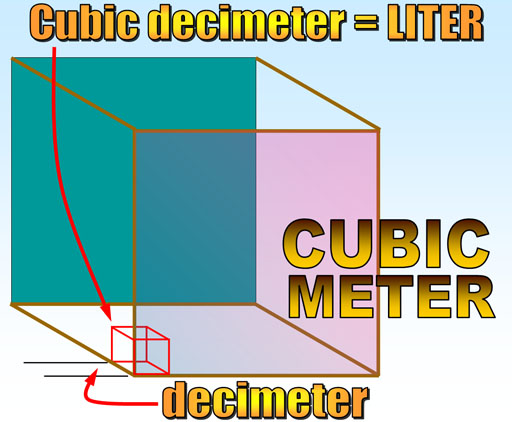
When the metric system was created, it cleverly set a certain volume of water to be the standard for mass.
The meter was the standard for distance (distance from equator to North Pole divided by 10 million). A standardized volume (more specifically a cubic meter) can be made by making a box with one meter on all sides. However, a cubic meter is a pretty large volume (bigger than a cubic yard). In the metric system, we size things either 10 times larger or 10 times smaller. So let's divide the meter by 10 to get a more useable volume. One meter divided by 10 is called a decimeter because deci means one tenth. Now if you make a cube that has a height, width, and depth of one decimeter, you create a cubic decimeter. That is a more convenient size, which is a little larger than a quart. Since this is a common size, the cubic decimeter was given a shorter name. The French called it the Liter after the word, Litron, an older measure for capacity.
The standard for mass came from filling one liter with pure water and its mass was called a kilogram (1000 grams).
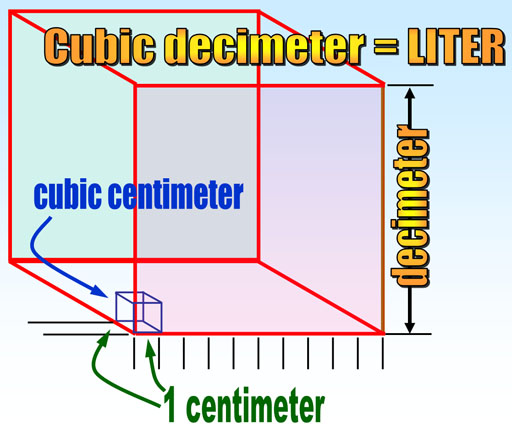
Cubic centimeter of water equals one gram:
It takes 1000 cubic centimeters to make one cubic decimeter (which is the same as one liter). That means one cubic centimeter is the same volume as 1/1000 of a liter. The French word of "milli" means 1/1000 (one thousandth). So on cubic centimeter is the same size as one milliliter. That also means that one cubic centimeter (or milliliter) of water weighs 1 gram.
In the English system, there is no easy connection between the volume of water and the weight of water. For example, one quart of water does not weigh one pound. Fortunately, in the Metric system, if you know the volume of water, you can figure the mass of water in your head.

The Aquafina water bottle holds one half liter (500 milliliters) of water. So you know it weighs close to 0.5 kilogram (or 500 grams). Since a kilogram is about 2.2 pounds, half a kilogram would be 1.1 pounds.
The 12 pack of half liter water bottles would be 12 x 0.5 kilograms (6 kilograms). 6 kilograms x 2.2 lbs per kilogram = 13.2 lbs plus weight of the plastic bottle containers. So even before picking one of these 12 packs up, you can calculate that it's about the weight of a bowling ball.

Americans really need to think in metrics rather than in English units. The rest of the world plus nearly all scientific disciplines in the USA have changed to the Metric system. So when you pick up an item that shows mass or volume, get in the habit of looking at both the metric and English units on the label. For example, a 12 ounce can of soda is listed as 355 mL (milliliters). The advantage of milliliters is that water weighs essentially 1 gram per milliliter. So this can of diet soda weighs around 355 grams. If you only looked at 12 fluid ounces, the mass of the can would not be easy to figure out.

Water has a tendency to creep up the inside wall of the graduated cylinder. So the water surface isn't flat. It will be bowed. They call this curved surface the meniscus from the Greek word meniskos meaning "crescent" like the crescent of the moon. The bottom of the curve is where you read the water's volume. In the upper-right closeup image, the bottom of the meniscus is between 20.6 and 20.8 mL (The divisions here are 0.2 mL apart.) When you have your eye at the correct level, the lines on the graduated cylinder will look horizontal rather than curved. Notice the lines on the close-up image are horizontal. That means you have your eyes level with that area of the graduated cylinder.

What you are going to do is measure out a certain volume of water and then check how much it weighs. First of all, you need to find the mass of the 100 mL graduated cylinder when it's empty.
1) When you turn on the balance, wait until it shows "0.00" and the "g" is above it. Place the 100 mL graduated cylinder on the balance and write down its mass in grams. You will need the mass of the empty 100 mL graduated cylinder later. (Note: Any amounts you write down, will be included in the lab report)
2) Fill the graduated cylinder to the 50 mL mark using tap water. If you add too much water, pour some out until you get as close as you can to the 50 mL mark.
3) Dry off any water that may have gotten on the outside of the graduated cylinder. We only want the mass of the 50 mL of water on the inside.
4) Turn on the balance again and wait for "0.00". The set the graduated cylinder with the 50 mL of water onto the balance. Write down the mass in grams.
Mass of graduated cylinder with water
minus
Mass of empty graduated cylinder
equals
Mass of just the water.
5) Calculate the mass of the water by subtracting the mass of the empty graduated cylinder from the mass of the cylinder with 50 mL of water in it.
A1) What is the mass of 50 mL of water? It should be close to 50 grams plus or minus a few grams. Be sure to report out to the hundredths place (all digits shown).

6) Now add more water until you get to the 100 mL mark. Dry off any water on the outside of the cylinder.
7) Turn on balance again and wait for "0.00". Record the mass shown.
8) Calculate the mass of the water by subtracting the mass of the empty graduated cylinder from the mass of the cylinder with 100 mL of water in it.
A2) What is the mass of 100 mL of water? It should be close to 100 grams plus or minus a few grams.

I've seen cookbooks that substitute Dr. Pepper (or other non-diet sodas) for sugar. One reason is that sodas have a lot of sugar in them.

On the can under "Nutrition Facts" you can see that this can of Dr. Pepper has 40 grams of sugar. Unfortunately, most recipes don't list grams of sugar but the volume of sugar in tablespoons or cups. So your task will be to weigh out 40 grams of granulated sugar and place it into a 100 mL graduated cylinder that will show the volume.




3) Place the weighing boat onto the balance before you turn it on. When it comes on, it will automatically TARE (throw away) the mass of the weighing boat and show "0.00" g.
4) Orient the weighing boat so that you can see the digital screen.

5) You can start pouring out sugar onto the weighing boat while watching the digital readout of the balance. Remember you are trying to weigh out 40 grams of sugar, which is the same that's in most 12 ounce non-diet sodas.
6) Tapping on the box of sugar to help the sugar come out more evenly. If you are using a sugar bowl, then you can use a spoon to get sugar onto the weighting boat.
If like me, you will be surprised how much sugar is in just one 12 ounce can of soda.
If you interrupt pouring for a minute, your balance will shut off and you will have to place the sugar in some other container and start over.


7) Now with the aid of the weighing boat you can pour the sugar into the 100 mL graduated cylinder. It may be helpful to hold the graduated cylinder and tilt it.
8) Tap on the weighing boat to help the sugar pour more evenly.
9) After all the sugar is in the 100 mL graduated cylinder, set the graduated cylinder on a flat surface and tap it so the sugar settles. I got 44 mL volume for that 40 grams of sugar.
B1) What volume of sugar did you get when you weighed out 40 grams of sugar?

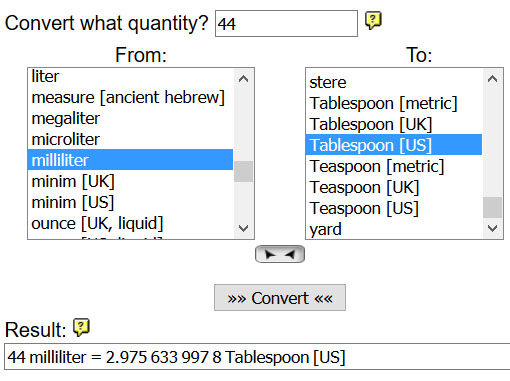

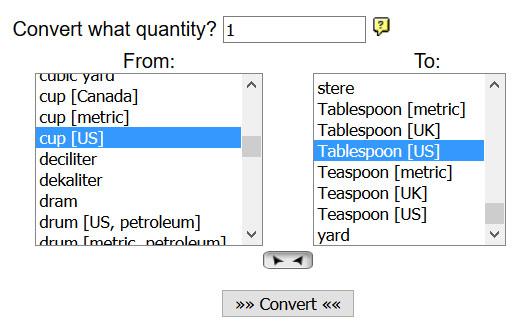
10) Go to the onlineconversion.com website and find the Volume link (near bottom of the page). Set the conversion to have a quantity of "1" and the "From:" unit as "cup [US]" and the "To:" unit as "Tablespoon [US]". Click the "Convert" button to see how many Tablespoons are in a cup.
B2) How many tablespoons are equal to one cup?

11) A Double Big Gulp drink is 64 oz. We know that 12 oz of a non-diet soda contains about 3 tablespoons of sugar (3 tablespoons per 12 oz). To figure out how many tablespoons of sugar in 64 oz, we can use the below calculation because it is set up with tablespoons on top (numerator) and the ounces will cancel leaving just tablespoons as the answer.
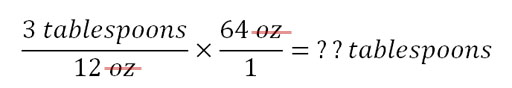
B3) How many tablespoons of sugar is in a Double Big Gulp drink?
B4) Would drinking a Double Big Gulp give you the same sugar as eating one cup of sugar?


Candy manufacturers are required to print the net weight of the candy bar on the label. "Net weight" means the weight of just the candy bar without the wrapping. The weight will be shown in ounces and grams.
1) Get a candy bar or bag of chips (if you prefer something healthier, that's fine too as long as it is packaged with weight and barcode).
C1) What is name of the candy bar or the bag of chips?
C2) How many grams does it say the candy or chips weigh?

2) On the back of the wrapper you should see the UPC (Universal Product Code). It will be two single digit numbers on both ends (0 & 4 here) and two 5-digit numbers in the middle. This candy bar has "1 28000 20660 4" as its UPC. The left "28000" is usually the code for a particular manufacture (Nestle in this case). The "20660" is the code for this particular product (100 Grand Share Pack).
C3) What is the UPC for your candy bar or bag of chips?

3) Turn on the scale and be sure it says 0.00 with "g" for grams at the top.
4) Place the wrapped candy bar or bag of chips on the scale. If they block the readout window, place a cup or something on the scale to raise up the candy bar or chips. Press TARE to zero out the weight of whatever was used to raise it. (This is similar to what you did when weighing an envelope.). Now weigh the candy bar or chips.
C4) What is the weight of the candy bar or chips with its wrapper?
5) Eat the candy bar or chips (or give it to someone), then weigh just the wrapper.
C5) What is the weight of the wrapper?
6) Subtract the weight of the wrapper from the weight in weight of the candy bar or bag of chips with their wrapper.
C6) What is the weight of just the candy bar or the chips (without their wrapper)?
Notice if your weight (mass) is close to what the manufacturer says it is. There have been cases where manufacturers try to save money by selling products that have less weight or volume than what they list on the package.
If you wish, you can copy the below summary into your email (or Word document) and type your answers after the descriptions. Then email them to Ken Costello at chm107@chemistryland.com. Make sure the Subject line of the email says "Field Lab 2"
A) Experiment confirming one milliliter of water weighs one gram
A1) What is the mass of 50 mL of water?
A2) What is the mass of 100 mL of water?
B) Determining volume of sugar using its mass:
B1) What volume of sugar did you get when you weighed out 40 grams of sugar?
B2) How many tablespoons are equal to one cup?
B3) How many tablespoons of sugar is in a Double Big Gulp drink?
C) Confirming grams of a candy bar or bag of chips:
C1) What is name of the candy bar or the bag of chips?
C2) How many grams does it say the candy or chips weigh?
C3) What is the UPC for your candy bar or bag of chips?
C5) What is the weight of the wrapper?
C6) What is the weight of just the candy bar or the chips (without their wrapper)?