Solutions are important to chemistry because it is the best way to mix chemicals so that they are in contact with each other. That speeds up the reaction between the chemicals. In the lab that focused on double-replacement reactions, we first made a solution of each compound and then mixed those solutions. The reactions were immediate.
Also, most of the chemical reactions that make life occur in solution. So understanding the properties of solutions is quite useful.
1. Classify compounds strong electrolytes, weak electrolytes, or non-electrolytes by their conductivity.
2. Observe Heat of Solution.
3. Observe freezing point depression, which is a colligative property of solutions.
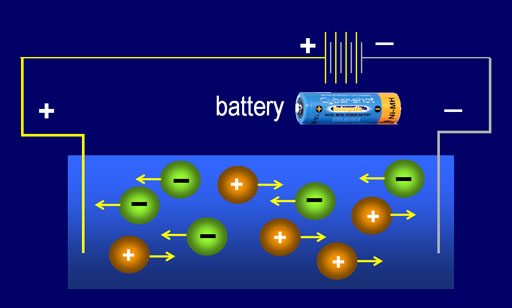
An electrolyte is a substance that produces ions in a solution. Because of those ions, the solution of an electrolyte has electrical conductivity. Remember, the TDS meter is based on ions conducting electrical current.
An electrolyte can be classified as strong or weak depending on how well it conducts electricity. Strong electrolytes exist as positive or negative ions (not uncharged molecules) in water. For example, in a water solution, strong acids are completely ionized:
HCl(g) → H+(aq) + Cl-(aq)

Weak electrolytes do not completely ionize. In other words, most of the weak electrolyte dissolves without dissociating into ions. The fraction that does ionize allows for low electrical conductivity and therefore is a weak electrolyte. Acetic acid is a good example of a weak electrolyte. The large arrow says the reaction mostly goes towards producing the acetic acid molecule. The small arrow means some of those acetic acid molecules dissociate into ions (hydrogen ions and the acetate ions).
CH3COOH(aq) ![]() H+(aq) + CH3COO-(aq)
H+(aq) + CH3COO-(aq)

Nonelectrolytes dissolve in water but remain as intact molecules that do not dissociate into ions. Dissolved sugars (like syrups) are good examples of nonelectrolytes. Below is equation using glucose. Again, sugar dissolved in pure water does not conduct electricity.
C6H12O6(s) → C6H12O6(aq)
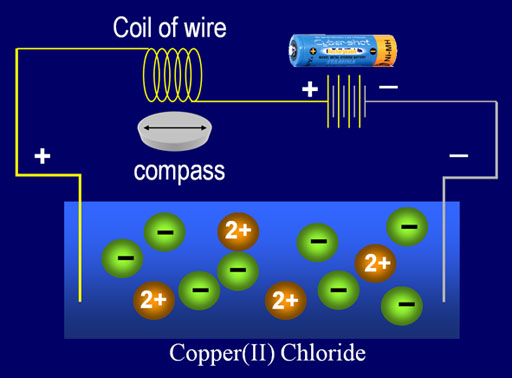
"-lyte" also indicates that this is the substance that can undergo "Electrolysis". "Lysis" means about the same thing as lyte. It is the pulling apart or decomposing of something. Electrolysis means the breaking apart of something using electricity. Electrolytes are vulnerable to electrolysis. Put them in water and attach wires from a 9 volt battery and they will decompose. Copper(II) chloride (CuCl2) put in water will dissolve into copper ions and chloride ions. Let the water evaporate and you get back the copper chloride. However, expose it to a battery and the negative chlorine ions will be attracted to the positive side of the battery, and the ions turn into chlorine gas and bubble away. The positive copper ions will be attracted to the negative side of the battery and will plate out on that wire as copper metal. As this happens the copper chloride is "decomposed."

In this lab we are not really decomposing the electrolytes using electrolysis. We are just dissolving them in water. (Not as much fun but easier to do). If a substance dissolves in water, it gives you insight as to the make up of the that substance. In other words it tells you that there are charges or partial charges on the substance that water is pulling on. If the solution conducts electricity, it tells you more. It says that the substance has split apart into ions.
Remember chemistry has the disadvantage of trying to study things that are too small to see. So we depend on these indirect methods of "looking at" the substances. For example, when the TDS meter gives a high reading when table salt (NaCl) is dissolved in water, we can "visualize" the sodium ions (Na+) moving independent of the chloride ions (Cl-).

We have all seen these warning signs on items like hair blow dryers. We all know that dropping an electrical device in water is risking electrocution. However, it's not water that causes the electric shock. It's the ions from impurities in the water that allow electricity to flow into your body or hand. In a bathtub of pure water, you would be safe if an electric blow dryer fell into the water because pure water is a non-electrolyte However, tap water is not safe because of the ions from dissolved salts, which are electrolytes.
In the chart below, the strong electrolyte examples will completely ionize (completely dissociate into ions). In the case of the strong acids, one ion will always be the hydrogen ion (H+). The weak electrolytes will also dissolve in water, but only a small fraction of electrolyte splits apart (dissociates) into ions. The nonelectrolytes dissolve in water, but essentially none of the molecules dissociate into ions.
Strong Electrolytes |
Weak Electrolytes |
Nonelectrolytes |
| Strong acids: Sulfuric acid (H2SO4), nitric acid (HNO3), hydrochloric acid (HCl), hydrobromic acid (HBr), perchloric acid (HClO4) | Weak acids: Acetic acid (CH3COOH), carbonic acid (H2CO3), nitrous acid (HNO2), sulfurous acid (H2SO3), hydrosulfuric acid (H2S), oxalic acid (HOOCCOOH), boric acid (H3BO3), hypochlorous acid (HClO), and hydrofluoric acid (HF). | Molecules: H2O, sugar, methanol (CH3OH), ethanol (CH3CH2OH) |
| Strong bases: sodium hydroxide (NaOH), potassium hydroxide (KOH), calcium hydroxide (Ca(OH)2), barium hydroxide (Ba(OH)2). | Weak base: NH3 NH3 + H2O → NH4+ + OH- |
|
| Most soluble salts: Sodium chloride (NaCl), lithium chloride (LiCl), copper(II) chloride, sodium carbonate (Na2CO3). |


Haz Mat Application:
This image shows power lines down on an asphalt road in the rain. Rain is pure water which does not conduct electricity. Asphalt has no electrolytes in it. So electrocution in this situation is not as likely. However, if a person steps onto the dirt or grassy area, the electrolytes (salts) in the soil will conduct electricity into the person's body.
Lesson: Stay on asphalt and only walk through freshly fallen rain water. If the water is muddy, then it has electrolytes in it, which means it will conduct electricity. Of course at very high voltages, even areas without electrolytes can still result in electrocution.

Battery Application:
In the table above, potassium hydroxide (KOH) is listed as a strong electrolyte, which means it is good at conducting electricity. Potassium hydroxide is used to make alkaline batteries, which get their name from the alkaline potassium hydroxide used in the batteries.

Eye care application:
Boric acid (H3BO3) is a weak electrolyte, which means very few of the acidic hydrogen ions split away from the boric acid molecule. This makes solutions of boric acid very weak acids. Boric acid is anti-fungal and anti-bacterial, so it is good for eye infections, and because it is such a weak electrolyte, it is safe in eyewashes.

Forensic application:
When crime labs receive white powders as evidence, they try to identify the powder. If the powder dissolves, it means the unknown substance is polar or ionic. If the solution is very conductive (reads high on TDS meter), then that means it is ionic and a strong electrolyte. This helps narrow the list of possible chemicals or drugs. For example, some drugs are treated with hydrochloric acid (a strong electrolyte) to create a hydrochloride salt of the drug. This makes those drugs more soluble and strong electrolytes. The image is of cocaine hydrochloride solution. That solution would read high on the TDS meter. If not, then someone substituted the cocaine hydrochloride with something else.

Health Application: The body loses electrolytes during times of profuse sweating, diarrhea, vomiting, or through excessive retention or elimination of urine. Electrolytes help the muscles contract, move nutrients in and out of the cells, assists in nerve and brain functions, plus many other functions.
The electrolytes used in the body are sodium (Na+), potassium (K+), calcium (Ca2+), magnesium (Mg2+), chloride (Cl-), hydrogen phosphate (HPO42-), and hydrogen carbonate (HCO3-). Sodium, potassium, and calcium are the ones that are more apt to be out of balance. The electrolyte salts of sodium chloride, potassium chloride, and calcium chloride, and a calcium carbonate are the usual sources of these electrolytes.
Attractions broken: ions are separated |
|
NaOH(s) → Na+ + OH- |
|
Attractions broken: Water molecules are separated |
|
H2O–H2O–H2O → H2O + H2O + H2O |
|
Attractions formed: Water attracted to ions |
|
H2O | H2O – Na+ – H2O | H2O |
H2O | H2O – OH- – H2O | H2O |
When gases, liquids, or solids dissolve in a solvent (like water), heat may be generated or absorbed. It depends on the total energy of the three below steps. Let's use sodium hydroxide (NaOH) as the example.
Attractions Broken: To get NaOH to dissolve in water, the ionic bonds between the sodium ions and the hydroxide ions have to be broken. That takes some energy, which means this step is endothermic (absorbs energy).
Attractions Broken: Before NaOH is added, the water molecules are held together by their partial charges and from hydrogen bonds. For Na and OH ions to mix with the water, the water molecules have to be split apart. So that takes some energy to break those attractions. This step is endothermic.
Attractions Formed: As the sodium ions come close to the water molecules, the water molecules race towards the positive sodium ions because the negative side of water is attracted to a positive charge. As the water molecules race towards the sodium ions, they run into other water molecules which releases heat energy. The same is true with the OH- ions. The positive sides of other water molecules will race toward the OH- ion. Energy is released as they come run into other water molecules or come to a stop next to the OH- ion. This step is exothermic.
Attractions formed: The partially (δ) negative side of water molecules are attracted to the positive sodium ion.
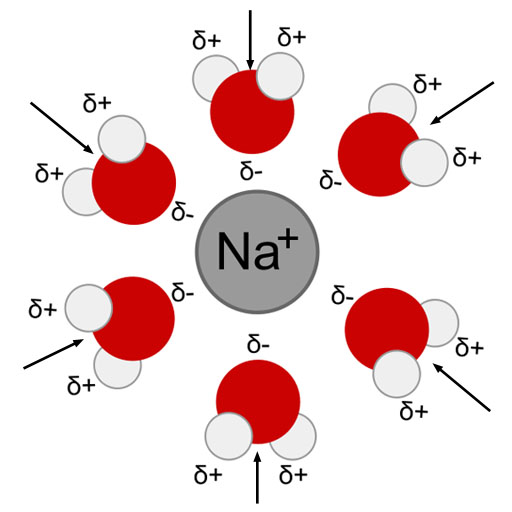
Heat of Solution then depends on how much energy each of these steps either require or release. When things are attracted to each other, it takes energy to pull them apart. So the first two steps require energy. When things that attract each other are allowed to come together, they will release energy as they collide with things along the way or at the point they collide with what they were attracted to. Think of two magnets that fly towards each other because of the magnetic attraction. When they slam into each other, energy in the form of sound waves is released along with some heating up of the magnets themselves. The heat isn't very noticeable, but it's there.
If the energy needed to break those attractions in the first two steps is more than the energy released in the last step, then the dissolving of NaOH will cause the water to get colder. However, if the last step produces more energy than the first two steps, then the water will get hotter when NaOH dissolves. If tested you will find that when NaOH is dissolved in water, the water gets hotter. So the step where the H2O molecules race towards the Na+ ions and the OH- ions must produce more energy than the first two steps.

First Aid Application of Heat of Solution:
Some instant cold packs and instant heat packs use chemicals that upon dissolving their Heat of Solution absorbs energy (cold pack) or produces energy (hot pack).

Biodiesel Production:
The first chemical reaction done in the production of biodiesel is the dissolving of sodium hydroxide in methanol. The Heat of Solution of NaOH in methanol makes the solution quite hot. Some plastic containers are softened by the heat, so that has to be avoided. Also, if rubbing alcohol is used by mistake, its Heat of Solution produces no heat. That's a sign that the wrong alcohol was used.
The middle and right bottles contain 100% methanol. The left bottle contains 100% isopropyl alcohol, which doesn't work in making biodiesel.

Acid Handling Safety:
The motto in chemistry labs is to "Never add water to acid; add acid to water." That's because the Heat of Solution of acid dissolving into water produces so much heat that the water can turn to steam and splatter the solution. If acid is added to water, the liquid that splatters will be mostly water. If water is added to acid, the liquid that splatters will be mostly acid. Also, water has a high ability to absorb heat. So adding acid to water will be cooled by the extra water.
If you want to be crazy, then do the opposite.

De-Icing Application:
All salts are good for melting ice because they reduce the melting point of ice. The salts that have a Heat of Solution that produces heat are even better because of the extra heat they produce as they dissolve into the melting ice. Calcium chloride is one of the electrolytes that has a Heat of Solution that produces heat. It's also less detrimental to the environment and to automobiles, so it is often used to de-ice roads.

An ice storm is coming. Your car’s radiator has no antifreeze in it; all stores are closed, and you’ve got to use whatever you have around the house to keep the water from freezing in the car’s engine and splitting the engine block.
Rule one: What you add has to dissolve in water.
Rule two: If you add a solid or liquid that dissolves in water, it doesn’t matter what it is, just the amount.
Rule three: Just like antifreeze, your goal is to replace about ½ of the water with a solid or liquid that is miscible with water.
Rule four: It is generally best if the substance isn't volatile (doesn't evaporate easily).

Examples 1: Sugar, salt, baking soda, shampoo, laundry detergent, pancake syrup.
Examples 2: Rubbing alcohol, brake fluid
The items on the left are what you had. So you mixed all of these into a gallon of water and poured it into your radiator.
Note: This is where it doesn't matter what substance you use. It's the count (the moles) that matters, not its chemical makeup.
It was a long cold night and the next morning you wonder if what you did saved you thousands of dollars on a new engine.


Freezing point depression
Reason 1: The addition of these other substances reduces the number of water molecules that are available to freeze.
Reason 2: By adding these other substances, you’ve added disorder to the mixture. Nature tends to favor disorder (entropy). When water tries to freeze, it has to get organized, which will be more difficult because it’s mixed with all of these other molecules.

This frog is frozen but is still alive because the water in it did not turn into ice crystals, which would have ruptured the cells in its body. Why didn’t ice crystals form?
Glucose and glycerin in its blood and cells prevent water from freezing. Glucose and glycerin dilutes the water, so that reduces the number of water molecules available to make ice crystals. Also, their presence created disorder in the water. So water as to overcome that disorder in order to arrange itself neatly into ice crystals.

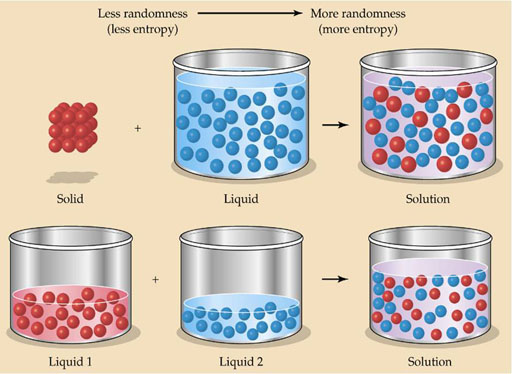
Pure substances are more orderly. When mixed, they lose that order and become more random (more entropy). Again, nature favors entropy.


Freezing Point Depression and Boiling Point Elevation
Antifreeze is also called a “coolant” because it can elevate the boiling point of water.
So, if you don’t have any antifreeze (coolant), what could you use instead?
Yes, the same items that you picked to keep it from freezing.

Let's talk more about freezing point depression.
Notice that ice cream melts differently than ice. Ice stays hard until it melts. Ice cream gradually get softer and softer.
Ice is a pure substance but ice cream is a mixture. In other words, the concentration of water in ice is 100%, but the concentration of water in the mixture is much less. So the 2-way movement of liquid water to frozen water favors frozen water because there are more molecules of water in the ice than out in the solution. So you have get colder than 0°C to get it to freeze.
About 30% of the water in ice cream never freezes because of the high level of dissolved solids like sugar, fats, and proteins.


Pure molten silica (SiO2) freezes into quartz at around 2000°C, but if mixed with CaO and Na2CO3, it freezes at about 1000°C. Other additives can bring it down to 500°C. Actually, at room temperature it is still not completely frozen. Glass is classified as a supercooled liquid.

Eggs are mostly water, but dissolved proteins keep them from freezing at 0°C.
Chefs take advantage of this in frozen desserts.

A story from Ken Costello:
At my office at the college, I stored sodas in a small refrigerator. One day I noticed that the Diet Coke had exploded after freezing, but the regular Coke did not freeze and therefore did not explode. I thought, "Why did the Diet Coke freeze and not regular Coke?" Regular coke has about 39 grams of sugar dissolved in it. Diet Coke has only about 0.1 grams of Aspartame sweetener in it. The 39 grams of sugar reduces the concentration of water. So the freezing point of regular Coke was lower than that of Diet Coke. Diet Coke's freezing point is basically the same as water since very little is dissolved in Diet Coke. So at 32°F, Diet Coke froze, but regular Coke did not.

The categories of strong, weak, and nonelectrolytes places substances into 3 fixed groups. In reality, there is a sliding scale from non-electrolyte to a strong electrolyte. Since electrolytes conduct electricity, we can use the TDS meter to measure how many ions are in solution and therefore reflect how good of an electrolyte a substance is.
This is the setup that we will use. The small 50mL beaker allows us to use only 20 mL of a solution to cover the probes of the TDS meter. The large beaker is for stability. The thermometer allows us to do a temperature adjustment on the TDS readings if we desire.

Let's begin with the liquid that is labeled "Pediatric Electrolyte". This is from a Walgreen's brand to compete with PediaSure's brand electrolytes.
Pour in at least 20 mL into the 50 mL beaker. You can pour all of it in if you wish.


Up close we can see the "x 10" readout. That means we are supposed to multiply the 254 by 10 to give us 2540 mg/L (ppm). That makes more sense. This is about 5 times the electrolyte (salt) level that is in Phoenix area water.
The temperature of 22°C is close enough to the standard 25°C that we won't bother doing the temperature adjustment on the TDS reading.
a1) What is the reading that you got with your Pediatric Electrolyte sample?
Take a photo of your setup for measuring the amount of electrolytes using the TDS meter.


We are now going to dissolve some compounds to see how good of an electrolyte they are depending on their TDS value. First you need to make sure your purified water is pure so that you know the conductivity is coming from your compound and not from your "pure" water. Here we see the TDS meter is reading 000 ppm for our purified water. If it is less than 10ppm, then it's OK.
a2) What is the reading of your purified water?


At first glance the reading looked like 146 ppm, but upon closer inspection we could see that the "x 10" multiplier was flashing. So that means the reading is 1460 ppm. Citric acid is considered a weak electrolyte, but it's stronger than other weak electrolytes.
a3) What is the reading of your solution of citric acid?
The electrical conductivity from the citric acid is from the two ions that it forms. The top molecule is the citric acid structure as a solid. In a water solution, the bottom structure shows the hydrogen ions that could be released leaving the negative citrate ion behind. Those hydrogen and citrate ions are responsible for conducting electricity.



Citric acid is what gives citrus fruits like oranges, lemons, and grapefruit their tart and sour taste. Food grade citric acid is also added to other foods to add tartness. If you wish, you can add a couple of scoops of this food grade citric acid to a glass from your kitchen and add a little water and taste it. Don't drink from one of the kit's beakers, those have been exposed to unsafe chemicals. Use a glass or cup from the kitchen.
a4) If you tried tasting a solution of citric acid, how did it taste?

Do the same thing with sodium chloride (NaCl). Add a little over 20 mL of purified water to the 50 mL beaker. Add a scoop or two of sodium chloride (from you kit or from the kitchen). Stir to dissolve the salt. Check it with the TDS meter.
In our sample, the reading was 586, but the "x 10" multiplier was flashing, so the reading is actually 5860 ppm (mg/L). This reading is higher than that of citric acid, which is expected because NaCl is a strong electrolyte, meaning essentially all of the NaCl will dissociate into Na+ and Cl- ions. Of course, to get a more accurate comparison between citric acid and sodium chloride, we would have to weigh the amounts we put into the water. For this experiment, we are just getting some rough comparisons.
The temperature of 22°C is close to the standard, so we aren't doing any temperature adjustment on the TDS reading for our purpose here.
a5) What is the reading of your solution of sodium chloride?


Now you will test acetic acid. This is a usual example of a weak electrolyte because less than 1% of the acetic acid actually has dissociated into H+ ions and acetate ions. Let's use the TDS meter to see how weak it is.
Your kit has about 9 mL of 5% w/v acetic acid (vinegar). You can pour that into the 50mL beaker. To cover the probes of the TDS meter, you need 20mL, so add purified water to bring it up to 20mL.

Since we had extra vinegar, so we didn't have to dilute it. Our reading was 757 ppm. If you used diluted vinegar, then your reading should be lower. The below reaction shows the ions that are responsible for the reading.
CH3COOH(aq) ![]() H+(aq) + CH3COO-(aq)
H+(aq) + CH3COO-(aq)
a6) What is the reading of your solution of acetic acid? Also, did you have to dilute it, or did you have some vinegar so that you didn't have to dilute it?
The TDS meter is mostly good at measuring the levels of strong electrolytes because it assumes that the electrolyte has completely ionized. Our reading of 757 mg per Liter is low because it is only reading the H+ and CH3COO-(acetate) ions, not all of the acetic acid in solution. Vinegar is 5% w/v meaning 5 grams per 100 mL. That converts to 50 grams per 1000mL (1 liter). The TDS meter only read 757 mg (0.757 grams) per liter instead of 50,000 mg (50 grams) per liter. "0.757 grams" divided by 50 grams is 0.015, so only 1.5% of the acetic acid actually dissociated into ions.

Put on goggles when working with this acid.

Now add 20mL of the 0.1M HCl to the beaker. Hydrochloric acid is a strong electrolyte. Let's see what kind of reading the TDS meter does with that.

The reading we got was 923 with the "x 10" multiplier. So that's 9230 mg/L. That's almost at the limit of the TDS meter.
a7) What is the reading of your solution of 0.1 M HCl?
The acetic acid above was 5% w/v, which is 5 grams/100mL, or 50 grams/Liter. Acetic acid weighs 60 grams per mole. Let's calculate how many moles per liter our acetic acid was.
50 grams x 1 mole =0.83 moles
1 Liter 60 grams 1 Liter
At 0.83 moles/L, that is 8.3 times more concentrated than the 0.1 moles/L for the HCl; however, the more dilute HCl solution showed a salt equivalent level of 9230 mg/L compared acetic acid of 757 mg/L. If HCl was at 0.83 moles/L instead of 0.1 moles/L, then its reading would have been 8.3 x 9230mg/L, which is 76609 mg/L. So at the same concentration, HCl has 101 times (76609/757=101)more ions in solution compared to acetic acid. So now we see why hydrochloric acid is called a strong electrolyte and acetic acid a weak electrolyte.

Add a couple of scoops of sugar to the 50 mL beaker and add a little more than 20 mL of purified water. Stir to dissolve the sugar.
Test the sugar solution with the TDS meter.
a8) What is the TDS reading of your solution of sugar?

Empty and clean the 50 mL beaker. Then add in a little over 20 mL of tap water. Our tap water read 468 ppm. The temperature was 20°C, so the actual reading is a little higher, but for the purpose of this next experiment, we won't make the adjustment.
a9) What is the TDS reading of your tap water (without any temperature adjustment)?

Take the 50 mL beaker with tap water out of the large beaker. Locate the test tube labeled "Ion Exchange Resin Dual Type".

Pour a few of the small beads from the test tube labeled "Ion Exchange Resin: Dual Type" onto the watch glass. Examine them with your mini microscope.
a10) What do the beads look like?


Now the tap water is retested. We found that the amount of electrolytes in the tap water has been lowered from 468ppm to 319ppm. So where did those ions go?
You may have noticed that this resin actually consists of two types of beads, light ones and dark ones. Below is a close up of those beads. The dark ones grab onto metal ions like sodium (Na+). The light beads grab onto the negative non-metal ions like chlorine (Cl-). The beads that grabbed the Na+ ions releases positive hydrogen (H+) ions. The beads that grab the negative ions such as Cl- release hydroxide ions (OH-). H+ and OH- then combine to make water (H2O). In other words, salt ions (one type of electrolytes) comes in, but they are replaced by the electrolytes of H+ and OH-, which combine to make pure water.

a11) What was the TDS reading after you added the ion exchange resin?
Take a photo of taking the TDS reading after adding ion exchange resin to tap water.
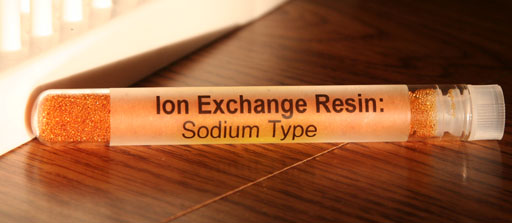
Extra Credit: Your kit also has some other ion exchange resin. This is the sodium type which is often used in water softeners. What these beads do is trap electrolytes such as Ca2+ and Mg2+ and replace them with sodium (Na+) ions. Calcium and magnesium ions latch onto soaps and cause soap scum. Hard water has high levels of calcium and magnesium ions. If those are replaced with sodium ions, it will be called soft water because sodium ions don't latch onto soap, so the soap works more efficiently. Place some of this resin in tap water and see if it changes the TDS levels.
a12) If you did this extra credit experiment, what was the TDS reading before and after you added the ion exchange resin: sodium type to tap water?

Round up these four compounds: Ammonium chloride (NH4Cl), potassium bromide (KBr), Citric acid, and lithium chloride (LiCl). You will be testing these for their "Heat of Solution". In other words, you will find out if heat is released or absorbed as the compound dissolves in water. For some chemicals, the temperature does not change as it dissolves.

Before we begin with lithium chloride, we need to know the temperature of the purified water that we will be adding to these compounds. So place the thermometer in a test tube and add about 2 mL of the purified water. That will be about 1 inch in depth.
b1) What is the temperature of the purified water to the nearest tenth of a degree Celsius?
Add 2 or 3 scoops of lithium chloride an empty test tube. In the image, that lithium chloride was added to the right test tube. See image for amount.

Add about 1 or 2 mL of purified water to the small amount of lithium chloride. That's about 1/2 inch to 1 inch depth

Move the thermometer over to the test tube with the small amount of lithium chloride and water. Use the thermometer to gently stir the mixture to help the lithium chloride dissolve. Feel the test tube where the dissolve lithium chloride is. It ought to feel hot.
Now check the temperature of the thermometer. If it is rising, wait until it reaches the maximum temperature. Because the temperature rose, the dissolving of lithium chloride is an exothermic reaction.
b2) What is the maximum temperature caused by the heat of solution of lithium chloride?

Now you will do the same thing with citric acid.
Now add 2 or 3 scoops of citric acid to a clean empty test tube. Then add 1 or 2 mL of purified water.
Use the thermometer to stir the solution. Then measure the temperature again to see if it rose, dropped, or stayed the same as the temperature of the purified water.
b3) What is the ending temperature caused by the heat of solution of citric acid?
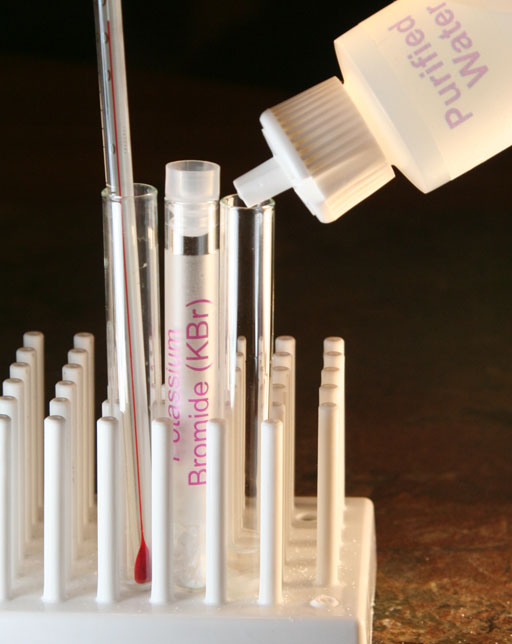
Now you will do the same thing with potassium bromide.
Now add a few crystals of potassium bromide to a clean empty test tube. Then add 1 or 2 mL of purified water.
Use the thermometer to stir the solution. Then measure the temperature again to see if it rose, dropped, or stayed the same.
b4) What is the ending temperature caused by the heat of solution of potassium bromide?

Now you will do the same thing with ammonium chloride.
Now add a 2 or 3 scoops of ammonium chloride to a clean empty test tube. Then add 1 or 2 mL of purified water.
Use the thermometer to stir the solution. Then measure the temperature again to see if it rose, dropped, or stayed the same. Also, feel the test tube where the solution is mixed to see if it feels colder or warmer than the other part of the test tube. If the temperature dropped, then the dissolving of ammonium chloride is an endothermic reaction.
b5) Does the bottom of the test tube feel colder, warmer, or the same as top of the test tube?
b6) What is the ending temperature caused by the heat of solution of ammonium chloride?
+14.8 kJ/mole

Round up the four chemicals shown: Copper(II) sulfate (CuSO4), potassium bromide (KBr), sodium chloride (NaCl), and lithium chloride (LiCl). Place them around the watch glass as shown.
In this experiment, we are not finding the exact freezing point depression caused by these chemicals, but we will observing their ability to cause a lowering of the melting point of ice (a freezing point depression).



You will notice that the crystal(s) will begin to dissolve into the ice pretty quickly.
c1) Does your crystal(s) of KBr start melting quickly into the ice cube?


Now place a small pile of sodium chloride onto the ice cube that was closest to where the sodium chloride test tube was laying. Notice what happens within a few seconds after placing some salt on the ice.
c2) Describe what you saw when you first placed the salt onto the ice.

Now place a small pile of lithium chloride onto the ice cube that was closest to where the lithium chloride test tube was laying. Notice what happens within a few seconds after placing some lithium chloride on the ice.
c3) Describe what you saw when you first placed the lithium chloride onto the ice.
Recall from the previous experiment that lithium chloride had quite a high heat of solution. So not only does it lower the melting point of ice like the other chemicals, it also produce quite a bit of heat when it dissolves, which helps with melting ice even more.

In a few minutes, the lithium chloride and sodium chloride have made small craters in their ice cubes. The potassium bromide crystal has melted into the ice and is now under the ice. The copper(II) sulfate crystal is sitting on the ice but has partially melted the ice below it.
We added more lithium chloride to the hole that it started and more sodium chloride to the hold it started. You should do the same to see if you can create a hole completely through the ice cubes.
Take a photo of the ice cubes after you have placed all four compounds on them.

On the left is a hole created by adding more lithium chloride. The addition of salt did a little more melting but our mixture of salt and water froze, so the melting more or less stopped.
c4) Describe what you are seeing regards to the melting of the ice due to adding more lithium chloride and sodium chloride.



If you wish, you can copy the below summary into your email (or Word document) and type your answers after the descriptions. The required photos can either be attached to the email or inserted in the Word document if going that route. Try to keep each image under 2 megabytes. If the first letter of your last name is between A and G, send your lab reports to Loree Cantrell-Briggs at Loree.Cantrell-Briggs@phoenixcollege.edu If the first letter of your last name is between H and Z, send your lab reports to Quinn Thacker at QRT2004@yahoo.com. Be sure to title the email "Lab 11".
a. Lab 11 Experiment 1: Conductivity of Electrolytes
a1) What is the reading that you got with your Pediatric Electrolyte sample?
Attach a photo of your setup for measuring the amount of electrolytes in the Pediatric Electrolyte solution using the TDS meter.
a2) What is the reading of your purified water?
a3) What is the reading of your solution of citric acid?
a4) If you tried tasting a solution of citric acid, how did it taste?
a5) What is the reading of your solution of sodium chloride?
a6) What is the reading of your solution of acetic acid? Also, did you have to dilute it, or did you have some vinegar so that you didn't have to dilute it?
a7) What is the reading of your solution of 0.1 M HCl?
a8) What is the TDS reading of your solution of sugar?
a9) What is the TDS reading of your tap water (without any temperature adjustment)?
a10) What do the beads look like?
a11) What was the TDS reading after you added the ion exchange resin?
Attach a photo of taking the TDS reading after adding ion exchange resin to tap water.
a12) If you did this extra credit experiment, what was the TDS reading before and after you added the ion exchange resin: sodium type to tap water?
b. Lab 11 Experiment 2: Heat of Solution
b1) What is the temperature of the purified water to the nearest tenth of a degree Celsius?
b2) What is the maximum temperature caused by the heat of solution of lithium chloride?
b3) What is the ending temperature caused by the heat of solution of citric acid?
b4) What is the ending temperature caused by the heat of solution of potassium bromide?
b5) Does the bottom of the test tube feel colder, warmer, or the same as top of the test tube?
b6) What is the ending temperature caused by the heat of solution of ammonium chloride?
c. Lab 11 Experiment 3: Freezing Point Depression
c1) Does your crystal(s) of KBr start melting quickly into the ice cube?
c2) Describe what you saw when you first placed the salt onto the ice.
c3) Describe what you saw when you first placed the lithium chloride onto the ice.
Attach a photo of the ice cubes after you have placed all four compounds on them.
c4) Describe what you are seeing regards to the melting of the ice due to adding more lithium chloride and sodium chloride.
