Carbon Dioxide: Experiment #4: Biodiesel.
Cleanup of raw biodiesel

Carbon dioxide is recognized as the biggest culprit to global warming. That's because the molecules of carbon dioxide let the visible light from the sun pass right through the atmosphere; however, when that visible light heats up the surface of the Earth, the Earth radiates out some of this heat as infrared light (infrared radiation). Carbon dioxide (like other greenhouse gases) will absorb infrared radiation and radiate half of it back towards the Earth.


So the carbon dioxide that comes from burning biodiesel is just carbon dioxide that was pulled from the air. So this carbon is being recycled. It is carbon from petroleum products that is adding extra carbon dioxide to the air that contributes to global warming.

Like Dissolves Like: This lab has to do with removing impurities from the biodiesel. To do that we are taking advantage of a basic chemistry principle called "Like Dissolves Like." In other words, things that are alike will mix with each other. If they are different, they won't. For example, if you don't have soap handy, you can use something greasy like vegetable oil or butter to clean greasy hands.
To learn more about "Like Dissolves Like," read the first section of my water tutorial at this link: WaterTutorial.htm

Visualizing Molecules:
The molecule here is methanol and is shown in
the "ball and stick" model format. It's called that because
it shows atoms as balls and the bonds that hold them
together as sticks. The four small white balls are hydrogen atoms.
The large gray ball is carbon. The red ball is oxygen. The balls that
represent the atoms are purposely made smaller so that you can see the
bonds between the atoms. The colors used are arbitrary not actual colors.
They are standard colors used for models.

Visualizing Molecules (cont'd):
Here is the methanol molecule again but using spheres that are more true to the size of the atoms. It is called the "spacefill" modeling format because the full space of the atoms are being represented. This is good but it hides the electrons being shared (bonds) that connect the atoms.
From the colors and sizes we can assume that we are seeing hydrogen (white), carbon (gray), and oxygen(red) atoms. What we can't see are the positive and negative regions that the molecule may possess.
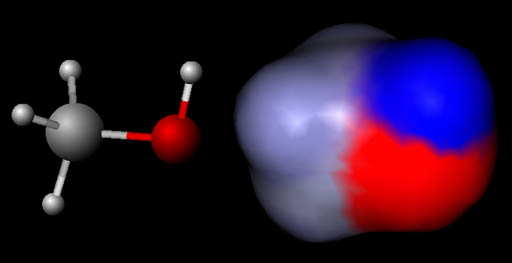
Visualizing positive & negative charges:
In this image the left molecule is methanol
shown in the "ball and stick" model like shown above. The right
image is also methanol but using something like the spacefill model above
except the colors are indicating regions of positive and negative charges.
The blue color means a positive region and the red region is a negative
region. The gray is neutral. The gray area is where the carbon and 3 hydrogen
atoms are. The red region is where the oxygen atom is. It is negative
because oxygen has a strong pull on electrons around it. The blue region
is where the single hydrogen atom is. It is positive because the oxygen
atom has pulled the hydrogen's electron away from it exposing the proton
which is in the center of hydrogen.
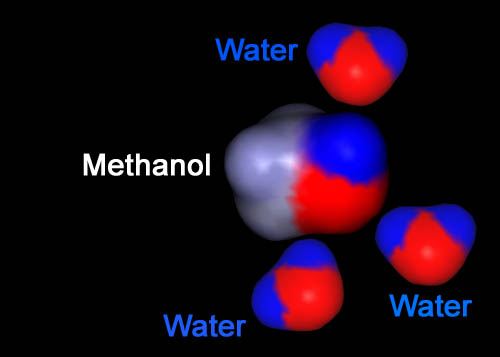
Just like the opposite poles of a magnet (north and south) attract, so does opposite charges (plus and minus). It is the opposite charges that causes molecules to be attracted to another. Here we see the negative (red) side of the top water being attracted to the positive (blue) region on the methanol. The bottom 2 waters have their positive (blue) sides attracted to the negative (red) part of methanol. This is why water dissolves (mixes with) alcohol.
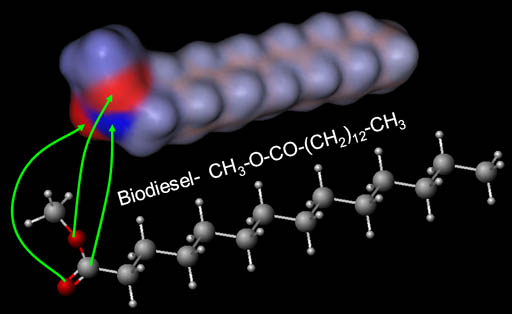
Biodiesel Mostly Neutral:
The end of the biodiesel that has the two oxygen atoms does have two negative (red) regions and one positive (blue) region; however, the majority of the biodiesel molecule is neutral (gray). That's why biodiesel does not dissolve in water.
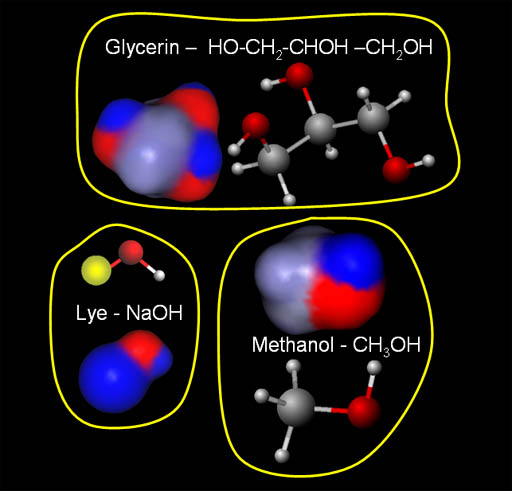
Contaminants in Biodiesel:
The molecules in the image are three of the main contaminants in the biodiesel. They are glycerin, sodium hydroxide (lye), and methanol. The good news is that each of them have negative and positive regions. That means water will be attracted to them and hold onto them. In other words, when the biodiesel is mixed with water, these contaminants will stay with the water when the water separates from the biodiesel.


Read full process of making Biodiesel:
It helps to see where the following lab fits into the
whole process. Before starting the lab on cleaning biodiesel, read my
tutorial on making biodiesel. Here is the URL:
SmallScaleBiodiesel.htm

The Biodiesel & Glycerin Vial:
You will find a vial with a metal screw cap labeled Biodiesel and Glycerin. These two products are the result of the molecules of vegetable oil being split apart. The glycerin sinks to the bottom.
Glycerin, by the way, is used in hand lotions and leather cleaners.

Shake the vial:
While sitting the biodiesel and glycerin in the vial has had time to separate, but you don't know how long that took. It's good to know about how long that times, so go ahead and shake the vial so the biodiesel and glycerin will be mixed.

Time How Long to Separate:
Like I said, it's good to know about
how long it takes for the biodiesel and glycerin to separate. The reaction
where vegetable oil is split into biodiesel and glycerin takes about 2
hours. The mixture is stirred the whole time. When stirring stops, you
wait for the biodiesel and glycerin to separate. Let's say you wait an
hour and nothing separates. Did that mean the reaction failed or you just
need to wait some more? 1. In your lab report, report about how long
it takes for the biodiesel and glycerin in your vial to separate (hint:
it's shorter than 20 minutes).

Wait for most of the glycerin to separate:
After the biodiesel and glycerin separates, you will still probably see some cloudiness in the upper layer (biodiesel). That's OK. It would take too long for that to clear up.

Transfer Glycerin to Test Tube:
Use the plastic pipette to draw the glycerin out of the vial and place it into an empty test tube.
You will probably want to hold the vial and tilt it slightly to make it easier to get all of the glycerin. I couldn't hold the vial because my right hand was busy holding the camera and taking the picture.

Transfer Glycerin to Test Tube:
Here I am placing the glycerin into an empty test tube. The clear liquid on top of the gold liquid is Biodiesel. You can draw some of that back into the pipette and put it back into the vial.
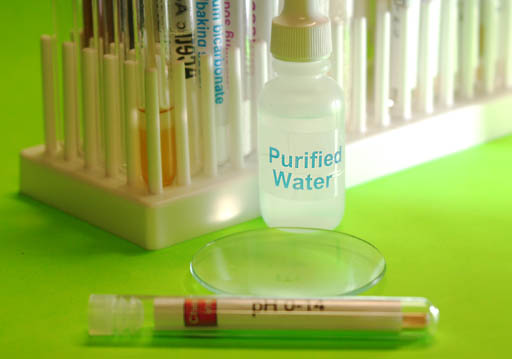
Test pH of Glycerin:
We want to test the pH of the glycerin. You should find the round watch glass in your kit. Also locate the pH paper that ranges from 0 to 14. From an earlier experiment, you probably have your purified water bottle filled with distilled water.
Use the same plastic pipette and draw out a little glycerin from the test tube and place 3 or 4 drops on the watch glass.
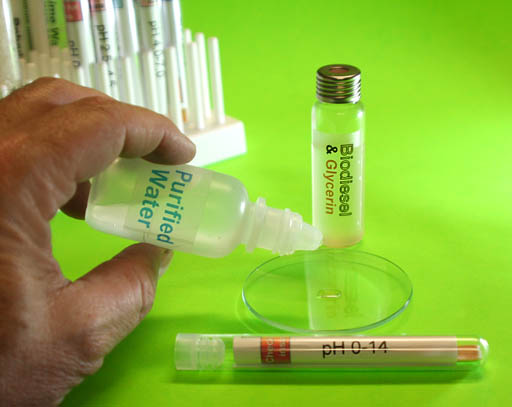
Test pH of Glycerin (cont'd):
After you have a few drops of the glycerin on the watch glass, place a few drops of the purified water on the glycerin.
The pH paper only works with aqueous (water) solutions.

Test pH of Glycerin (cont'd):
Before the 0-14 range test strip touches the liquid, it has 4 colors as shown. After it's wetted by the liquid, you see its new colors (inset).

Test pH of Glycerin (cont'd):
Compare the pH test strip to the various 4 color combinations that the test strip can have.
Here I found that it similar to either pH 11 or pH 12. That means the glycerin is alkaline (over 7 is alkaline). It is alkaline because some of the lye (sodium hydroxide, NaOH) used for the reaction is mixed with the glycerin.

Neutralizing Glycerin: Being alkaline, the glycerin (gold color liquid in the test tube) would not be good for your hands; however, acids could be used to neutralize the alkalinity. Remember acids have hydrogen ions (H+) and alkaline substances (also called bases) have hydroxide ions (OH-). When they come together in equal amounts they simply make water (H+ + OH- --> H2O). Therefore, adding just enough acid to neutralize the hydroxide in the glycerin would make the glycerin neutral. We aren't doing that in this experiment, but that's what would have to be done to make the glycerin usable in hand lotion.

Washing Biodiesel Layer:
Our attention now goes to the biodiesel remaining in the vial. This needs
cleaned before it can be used in an engine.
The biodiesel has residue of methanol, glycerin, sodium hydroxide (lye), and soap. All of these residues will dissolve in water. You might think that the distilled (purified) water would be good for washing; however, it tends to form an emulsion (a water and oil mixture that doesn't separate).

Tap Water is Best:
Water that has dissolved minerals (salts) does a better job. So the hard water we have in the valley is actually good for something. It's good for washing biodiesel.
Fill up your small (50mL) beaker with tap water..

Add Water to Biodiesel:
Pour the tap water into the biodiesel vial. Fill it to the neck of the vial. You need some air space so you can shake and mix it.

SHAKE:
Shake the vial to mix the biodiesel with the water. Shake for about 10 seconds.
 Take
a picture of this step, but show your face in the picture.
Take
a picture of this step, but show your face in the picture.

Time how long to separate:
Start your stop watch or look at a clock, so that you can time how long it takes for the water to separate from the biodiesel. Water is more dense than the biodiesel so it will be on the bottom.
2. Report this time in your lab report.

Add Salt if No Separation:
Sometimes the water and biodiesel forms an emulsion because of residue of soap. When the vegetable oil is in contact with the lye (NaOH), some of it is converted to soap. As you know soap will dissolve in both oily substances (like biodiesel) and water. In other words the soap allows the biodiesel to stay suspended in the water to form an emulsion. Salt added to the mixture will help break up the emulsion. It probably makes the soap less effective or it makes the water have more charges making it even more different than the biodiesel.

Checking pH of Wash Water:
After the water has settled to the bottom, use a clean plastic pipette to draw out a little water and place about 10 drops of water onto the watch glass. Make sure the watch glass has been wiped off from the previous step where the glycerin was placed there.

Checking pH of Wash Water (cont'd):
Use a new 0-14 pH strip paper, submerge it in the wash water.
 Take
a picture of this step.
Take
a picture of this step.

Checking pH of Wash Water (cont'd):
Check the paper against the 0-14 Chart. Here mine appears to have a pH between 9 and 10. That says the biodiesel was alkaline and not neutral (pH 7). Just like the glycerin, it seems the biodiesel has some sodium hydroxide (NaOH) residue. The wash water has pulled some of the NaOH away from the biodiesel because NaOH dissolves easily in water. Report what your pH is.

Dispose of Wash Water:
Draw the wash water out of the vial and dispose of it down the drain. The wash water contains a little soap and a little sodium hydroxide. This will not hurt the drain. Concentrated sodium hydroxide is what's used in drain cleaners like "Drano".

Washing Biodiesel Again:
Since our last wash water was alkaline, we need to repeat the washing until our pH paper indicates a pH of 7, which is neutral. Therefore, pour some more tap water into the biodiesel like before.

Washing Biodiesel Again (cont'd):
Like before, shake the biodiesel/water mixture for about 10 seconds and then let settle.
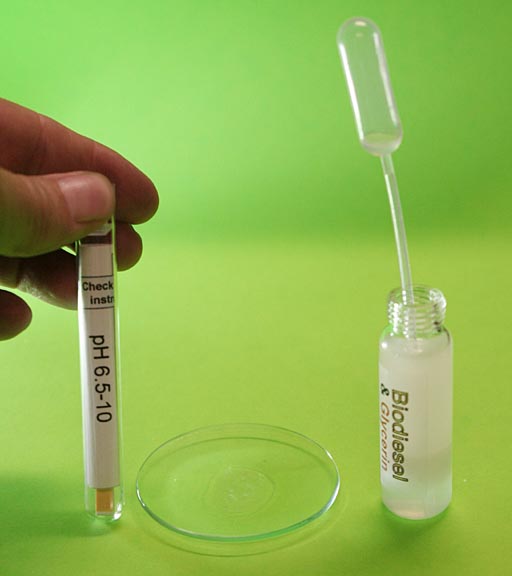
Checking pH of Wash Water #2:
Because we know the pH is somewhere between pH10 and pH 7, we can use the pH paper that is more accurate in this region. It is the pH 6.5-10 range paper.
Draw some wash water from the biodiesel and place on a cleaned watch glass. Note: If using the same plastic pipette, it would be a good idea to draw up some distilled water and rinse out the pipette first.

Checking pH of Wash Water #2 (cont'd):
Dip the 6.5-10 pH paper into the wash water.

Checking pH of Wash Water #2 (cont'd):
Compare the pH strip with the colors on the 6.5 - 10.0 range. Here mine looks like is close to 9.5. That means more sodium hydroxide was extracted from the biodiesel making the wash water alkaline.

Washing Biodiesel Third Time:
Dispose of any wash water that has settled in the vial and the then pour tap water a third time into the biodiesel. Shake it again and let it settle. Now we will check the pH again.

Checking pH of Wash Water #3:
Again we draw some wash water from the water layer of the vial and place on a clean watch glass. Again use the 6.5-10 pH paper to test it.
Notice the biodiesel label on the vial is coming off. That's because some biodiesel got on the label. Biodiesel is a great solvent that dissolves most anything. That's one of the uses I find for it.

Checking pH of Wash Water #3 (cont'd):
Compare the color of the pH strip to the colors on the 6.5-10.0 pH chart. Here it shows the wash water is neutral (around 7). That means we got all of the sodium hydroxide contaminant away from the biodiesel. (In your lab report tell me what was your final pH).
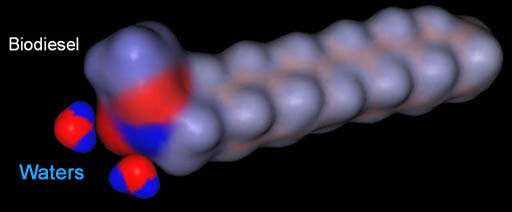
Water as new contaminant:
Even though washing with water removed the contaminants it introduced
a new one, which is water itself. Remember one end of the biodiesel has
charged regions which will attract some water. We need to remove this
water.
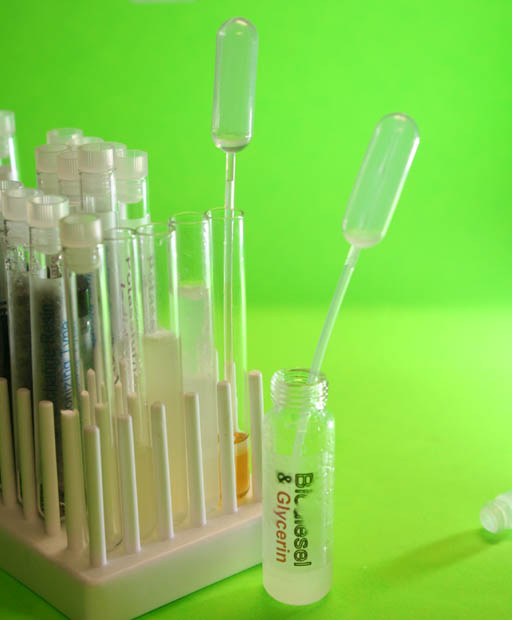
Transfer Biodiesel to Test Tubes:
Use a plastic pipette to transfer the biodiesel in the vial to two clean test tubes.
We are going to try out two methods of getting rid of the water that's in the biodiesel.

Transfer Biodiesel to Test Tubes (cont'd):
Fill the two test tubes with about the same amount of biodiesel. If need be transfer some from one test tube to the other.
I also have test tubes with waste wash water and the waste glycerin. The glycerin and wash water can be disposed of down the drain.
Our next task is the dry the biodiesel in the two test tubes using different methods.

Cotton as drying agent :
Find the cotton balls and pull off a wad of cotton that you will stuff down one of the test tubes. So the wad of cotton needs to be larger than the diameter of the test tube in order to have a wad of cotton will fit snuggly in the test tube.
So why is cotton useful for absorbing water? (see next panel)
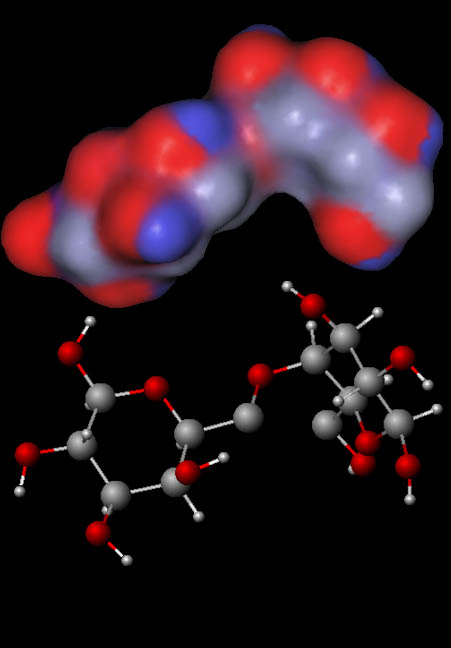
Cotton as drying agent (cont'd):
Cotton fibers are made of cellulose. Cellulose is chains of thousands of glucose molecules. This image is just two of the glucose molecules chained together. The images show both the stick and ball form of two glucose molecules and the charge distribution model.
With all of the oxygen atoms, we have several negative (red) regions. The hydrogen atoms attached to the oxygen are the positive (blue) regions. This means water is quite attracted to cellulose the same way water is attracted to sugar, which is why sugar dissolves.

To push down the cotton wad into the test tube, the rubber policeman in your kit would be handy for this.

Push Cotton Wad To Bottom:
Here I'm pushing down the cotton wad using the handle end of the rubber policeman. As you enter the biodiesel, push slowly giving time for the water in the biodiesel to be absorbed by the cotton fibers (cellulose).

Push Cotton Wad To Bottom (cont'd):
Here I have pushed the wad of cotton all the way to the bottom of the test tube.

Push Cotton Wad To Bottom (cont'd):
Notice how the cotton cleared up the biodiesel in the right test tube. The left test tube is where we will try a different method.

Drierite as drying agent:
A mineral called gypsum is used. Gypsum is the same mineral used to make dry wall, except the Drierite form of gypsum has had all water driven from it.
The mineral is calcium sulfate. Calcium sulfate is a salt, meaning that the calcium is positively charged and the sulfate in negative charged. That means water will be attracted to it.

Drierite as drying agent (cont'd):
This is the amount of drierite I want you to use, so just transfer that amount from your Drierite test tube into a clean test tube. This empty this amount of Drierite into the second test tube of biodiesel.

Drierite as drying agent (cont'd):
Pour the Drierite (calcium sulphate) into the second test tube of biodiesel.
Drierite as drying agent (cont'd):
Place the cap on the test tube and shake the biodiesel. This will allow the water in the biodiesel to come in contact with the Drierite, so the Drierite can absorb the water.

Pink indicates water was absorbed:
Drierite turns pinkish white when it has absorbed water. If all of the Drierite is pinkish white, then that means there still some water left in the biodiesel. Here I notice all of the Drierite is pinkish white. I decided to add a little more Drierite.
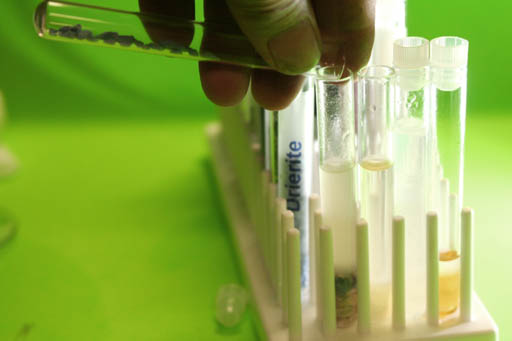
Add More Drierite if necessary:
You can add a little more of the Drierite to see if you can get all of the water out of the biodiesel.
After adding a little more Drierite, shake the test tube again.
 Take
a picture of this step.
Take
a picture of this step.

Add More Drierite if necessary (cont'd):
Here we see that a little bit of the Drierite is still blue. That means we got all of the water. I also dropped a few crystals of Drierite in the other test tube that had the cotton wad insert. Those crystals are still blue, so it helps confirm that the cotton got rid of all the water.
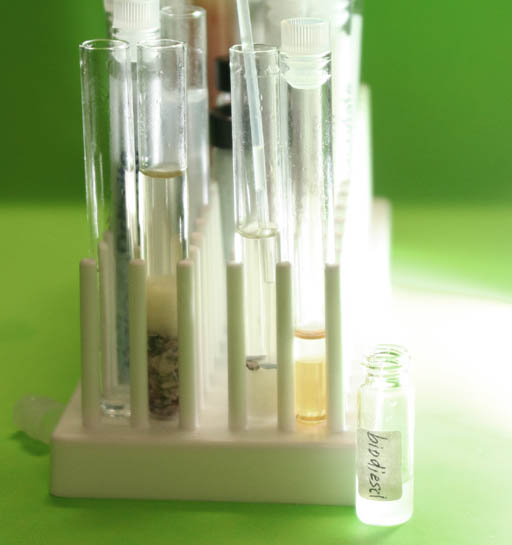
Save some of the biodiesel:
Choose which ever test tube that has the clearest biodiesel and transfer that to a small vial that you will find in the center of the test tube tray. Report which did the best for you. Was it the cotton ball or the Drierite.

Save some of the biodiesel (cont'd):
After filling the small vial, put the cap back on it. Save it.

Clean Up: Probably the most important part of this lab is to clean up all the items that got used in this lab. That would be 4 or 5 test tubes, plastic pipettes, a watch glass, a rubber policeman, and a beaker. Because biodiesel is oily, you will need some hot soapy water to get started. After soapy water, they all need rinsed with tap water following by a rinse with distilled water. A final rinse with rubbing alcohol is also recommended. For the plastic pipettes, you will need to draw (suck) cleaning liquids into them. Also, one test tube will have a wad of cotton at the bottom. Be careful in trying to get it out. The glass might break. If you can't get it out, just throw away the test tube.